Some Health Apps to be Regulated by the FDA
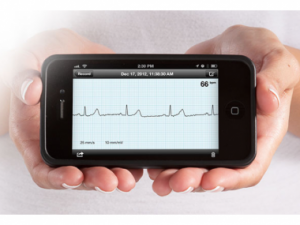 The New York Times (9/24, A12, Tavernise, Subscription Publication) reports that the Food and Drug Administration announced on Monday that “a small portion of the rapidly expanding universe of mobile health applications” would be regulated by the agency. FDA officials said the “goal is to oversee apps that function like medical devices, performing ultrasounds, for example, and that could potentially pose risks to patients.” Apps designed to work as pedometers and calorie tracking will not be regulated by the FDA, according to officials. Director of the FDA’s Center for Devices and Radiological Health, Dr. Jeffrey Shuren, said, “We have worked hard to strike the right balance, reviewing only the mobile apps that have the potential to harm consumers if they do not function properly.”
The New York Times (9/24, A12, Tavernise, Subscription Publication) reports that the Food and Drug Administration announced on Monday that “a small portion of the rapidly expanding universe of mobile health applications” would be regulated by the agency. FDA officials said the “goal is to oversee apps that function like medical devices, performing ultrasounds, for example, and that could potentially pose risks to patients.” Apps designed to work as pedometers and calorie tracking will not be regulated by the FDA, according to officials. Director of the FDA’s Center for Devices and Radiological Health, Dr. Jeffrey Shuren, said, “We have worked hard to strike the right balance, reviewing only the mobile apps that have the potential to harm consumers if they do not function properly.”
The Wall Street Journal (9/24, Burton, Subscription Publication) reports Shuren said, “We are only continuing our regulation for a small subset…of medical apps that pose a risk to patients,” for instance, apps that function as electrocardiograms.
The AP (9/24) reports that the small number of “apps that turn smartphones into devices, like a heart monitor, or medical attachments that plug into smartphones, like arm cuffs that measure blood pressure,” will be regulated. Shuren noted, “Mobile apps have the potential to transform health care by allowing doctors to diagnose patients with potentially life-threatening conditions outside of traditional health care settings, help consumers manage their own health and wellness, and also gain access to useful information whenever and wherever they need it.”
Also reporting on the story are CQ (9/24, Adams, Subscription Publication, 967), the AP (9/23, Alter), Reuters (9/24, Clarke), TheBaltimore Sun (9/23, Wells, Clarke), The San Jose (CA) Business Journal (9/23, Lynch, Subscription Publication),HealthDay (9/24, Reinberg), Medscape (9/24, Lowes, Subscription Publication), Modern Healthcare (9/23, Subscription Publication), and Healthcare IT News (9/24, Brino).

































