PharmaNest to Present Eight Digital Pathology and AI Abstracts at the International Liver Congress 2022
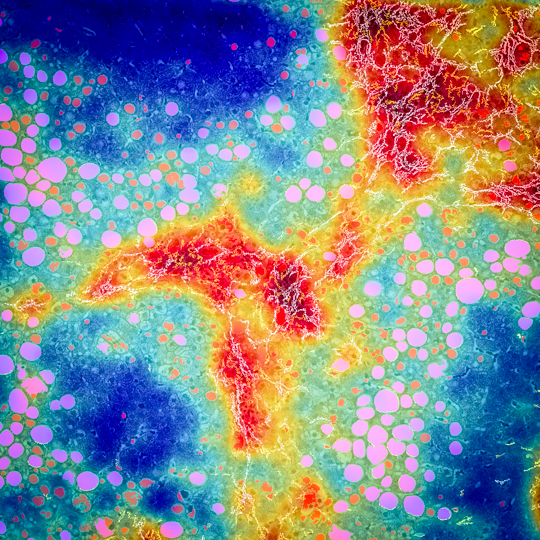
FibroNest generates augmented pathology images to visualize and quantify the histologic hallmarks of NASH
PharmaNest, an emerging leader in quantitative Digital Pathology and Artificial Intelligence, showcases the utility and performance of its FibroNest continuous score for the quantification of the severity of fibrosis and treatment effects of NASH therapies at the 2022 International Liver Conference.
PRINCETON, N.J., June 20, 2022 (GLOBE NEWSWIRE) — PharmaNest, an emerging leader in quantitative Digital Pathology and Artificial Intelligence, announces today that it will be present, alongside with its collaborators, eight presentations at this year’s International Liver Congress (ILC) in London, June 22-25, 2022. The abstracts highlight the utility and performance of FibroNest, the first multi-vendor quantitative digital Pathology image analysis and translational platform for the assessment of the severity and progression of Fibrosis and disease activity in NASH.
“The automated quantification of the phenotype of fibrosis severity from the same slides reviewed by pathologists offers a robust and scalable method to generate continuous scores with a detection threshold that can detect faint changes and drug effects,” said Mathieu Petitjean, CEO and founder of PharmaNest. Because the approach is fully quantitative and is not based on existing staging or grading paradigms, it can be used along the drug discovery and development process, and across etiologies of fibrosis.
The first abstract is a retrospective analysis of liver biopsies from the ARMOR Phase 2 trial of aramchol. It shows how Fibronest’s continuous fibrosis score detects drug effects that are missed by conventional histological methods (in partnership with Galmed Pharmaceuticals).
A second abstract includes results from a prospective analysis of liver biopsies from the FALCON1 Phase 2 trial of pegbelfermin. It demonstrates the sensitivity of FibroNest’s method to detect treatment and dose effects, with a performance that benchmarks imaging-based measurements (in partnership with Bristol Myers Squibb).
A third abstract discusses the performance of novel FibroNest computational methods for the continuous histological quantification of Ballooning and Inflammation in liver biopsies and related correlations with NASH-CRN histological grades.
Another abstract in collaboration with the University of Oxford uses FibroNest to develop an etiology-independent composite score for the automated histological assessment of fibrosis in liver biopsies.
Finally, several abstracts highlight the value of FibroNest to quantify the histological phenotype of fibrosis in the context of in-vitro assays for mid-throughput screening of antifibrotic compounds (in partnership with InSphero AG) and the validation for lead antifibrotic drug candidate in rodent models (in collaboration with Therapeutic Discovery department at Mount Sinai) with a performance that outperforms any alternative methods, when they exist.
The specific details of these abstracts are available on PharmaNest website at fibronest.com/science.
Media Contact:
Dr. Li Chen
Principal Investigator Contact
Li.chen@pharmanest.com
SOURCE: Globe Newswire
































